 |
By Yoskaly Lazo-Fernandez, PhD
The field of Cancer Stem Cell (CSC) research has been gaining traction in recent years1. CSCs are a minority group of cells (usually about 1 in 10000) within solid tumors of hematological cancers, which possess similar characteristics to normal stem cells. It is believed that these cells, which are identified by markers usually only found on tissue stem cells, are the only cancerous cells that can induce tumor development and propagation or metastasis2. Consequently, gaining a better understanding of the molecular machinery that originates and maintains CSCs is at the current forefront of the battle against cancer. Of particular importance is the identification of key molecular markers in CSCs, which may be targeted pharmacologically to disrupt their self-renewal capabilities.
A recent paper3 published in Nature appears to have identified several such targets. The Chinese group reported a mechanism of autophagy activation that induces CSC differentiation and proportion reduction. The final molecules that are inhibited in this signaling cascade are the transcriptional regulators: “inhibitor of DNA binding” isoforms 1 and 2 (ID-1 and -2). ID proteins (including 4 isoforms in humans) share one essential function, the inhibition of the basic helix-loop-helix (bHLH) transcription factors. The bHLH factors promote cell differentiation, thus their inhibition causes the opposite effect, increased cell proliferation and multipotency. As expected, ID protein expression is generally high in CSCs where they are essential for self-renewal and proliferation.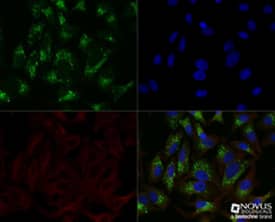 Immunocytochemistry/Immunofluorescence: p62/SQSTM1 Antibody [NBP1-48320] - HeLa cells were treated overnight with 50uM CQ, then fixed for 10 minutes using 10% formalin and then permeabilized for 5 minutes using 1X TBS + 0.5% Triton-X100. The cells were incubated with anti-p628/SQSTM1 at a 1:200 dilution overnight at 4C and detected with an anti-rabbit Dylight 488 (Green) at a 1:500 dilution. Alpha tubulin (DM1A) NB100-690 was used as a co-stain at a 1:1000 dilution and detected with an anti-mouse Dylight 550 (Red) at a 1:500 dilution. Nuclei were counterstained with DAPI (Blue). Cells were imaged using a 40X objective.
Immunocytochemistry/Immunofluorescence: p62/SQSTM1 Antibody [NBP1-48320] - HeLa cells were treated overnight with 50uM CQ, then fixed for 10 minutes using 10% formalin and then permeabilized for 5 minutes using 1X TBS + 0.5% Triton-X100. The cells were incubated with anti-p628/SQSTM1 at a 1:200 dilution overnight at 4C and detected with an anti-rabbit Dylight 488 (Green) at a 1:500 dilution. Alpha tubulin (DM1A) NB100-690 was used as a co-stain at a 1:1000 dilution and detected with an anti-mouse Dylight 550 (Red) at a 1:500 dilution. Nuclei were counterstained with DAPI (Blue). Cells were imaged using a 40X objective.
The mechanism, uncovered by Li et al.3 in their fascinating and well written article, begins with the activation of the calcium/calmodulin-dependent kinase II (CaMKII) by various calcium mobilizing agents, including vitamin D analogs. It has long been known that intracellular calcium ions are second messengers that participate in the regulation of many cellular functions including cell proliferation, and that intracellular calcium homeostasis is dysregulated in cancer cells4. It was also well known that vitamin D has cancer preventive properties5, and that its effects could be related to the activation of autophagy6,7.
Li et al3. then hypothesized and demonstrated that CaMKII directly phosphorylates Beclin 1, a key regulator of autophagy initiation, thus activating macroautophagy. In parallel CaMKII also phosphorylated ID-1 and ID-2, which promoted their K63-linked ubiquitination and p62-dependent transport to autolysosomes for degradation. The resulting ID-protein degradation stimulated the differentiation and reduced the proportion of CSC in neuroblastoma cell lines. This study provides robust experimental evidence of a complete cell signaling pathway that not only might explain the anticancer effects of vitamin D, but also establishes the basis for the design of new autophagy-related cancer therapies.Explore Autophagy Research Tools
 Yoskaly Lazo Fernandez, PhD
Yoskaly Lazo Fernandez, PhD
Emory University, Department of Medicine/Renal Division
Dr. Lazo-Fernandez is interested in understanding the dietary factors that contribute to the development of hypertension and other chronic diseases.
References